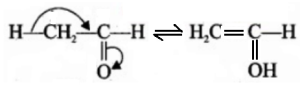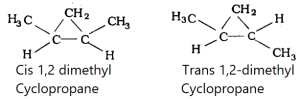Isomerism, Isomers and Their Properties
The compounds, generally, having same molecular formula but different structural formula are called isomers of each other. This phenomenon is called isomerism. They have different chemical and physical properties. As for example, ammonium cyanate (NH4CNO) and urea, CO(NH2)2 have same molecular formula and also have different chemical and physical properties. So they are isomers of each others.
Classification of Isomers:
Isomers of Organic compounds are of two types: (a)Structural Isomer and (b) stereo Isomer.
(a)Structural Isomers: The isomers differing in the arrangement of atoms or groups within the molecule are structural isomers. This phenomenon is structural isomerism. As for example, n-butane and iso-butane are structural isomers of each other.
n-Butane:CH3-CH2-CH3 ; iso-butane:CH3-CH(CH3)-CH3.
They are of five types- (i)Chain Isomer (ii)Functional Group Isomer (iii)Position Isomer (iv)Tautomer (v) Metamer.
(i)Chain Isomers: The isomers have the same molecular formula but differ in the order in which the carbon atoms are bonded to each other. This phenomenon is called Chain Isomerism. As for example, n-pentane, 2-methyl butane and 2, 2-dimethyl propane are chain isomers of each others.
(ii)Functional Group Isomers: The isomers have the same molecular formula but differ in their functional groups are called Functional Group Isomers of each other. This phenomenon is called Functional Group Isomerism. Dimethyl ether (CH3-O-CH3)and ethanol (CH3– CH2-OH)are Functional Group Isomers of each other.
(iii)Position Isomer: The position isomers are the isomers that can be formed by changing the position of the functional groups in their chains. This type of isomerism is called position isomerism. As for example, pentanone-2 (CH3– CO-CH2– CH2-CH3) and pentanone-3 (CH3– CH2-CO- CH2-CH3)are position isomers of each other.
(iv) Metamer and Metamerism: The isomers which have unequal carbon atoms on the two side of the functional group of organic compounds are called metamers. This type of isomerism is called metamerism. As for example, Propyl methyl ether (CH3-O-CH2-CH2-CH3) and diethyl ether (CH3– CH2-O- CH2-CH3) are metamers of each other.
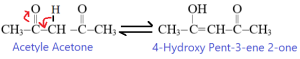
(v) Tautomer and Tautomerism: Special type of functional group isomers which are in dynamic equilibrium and can be converted into each other. The two different structures/isomers are called tautomers and the phenomenon is tautomerism. As for example, propanone (CH3-CO-CH3) and prop-1-ene 2-ol (CH3– C(OH)=CH2) are tautomers of each other.
This type of reaction is called ketone-enole tautomerism. Another example is
(b)Stereo Isomers and Stereo Isomerism:
The isomers differed in the relative arrangement in position of atoms or groups in three dimensional space within the molecule are known as Stereo Isomers and the phenomenon of stereo isomers is called Stereoisomerism. This types of isomers have the same molecular formula as well as same structural formula.
Classification of Stereoisomers: Stereoisomers are two types: (i)Geometric or cis-trans isomers (ii) Optical Isomer.
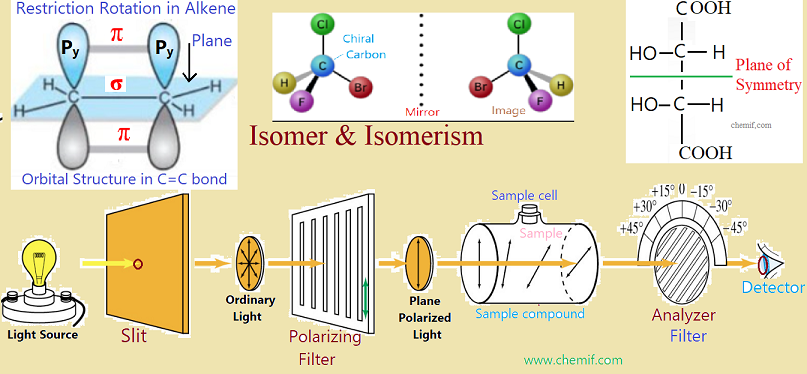
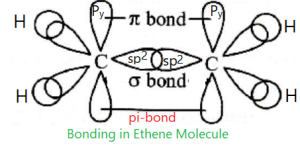
(i)Geometric Isomer: The isomers having the same molecular formula as well as same structural formula but differ in their geometric structure are cis-trans isomers or geometric isomers. This isomerism arises from a restriction in rotation about double bonds, or about single bonds in cyclic compounds. This type of geometric isomerism is usually observed in substituted alkene and cyclic compounds.
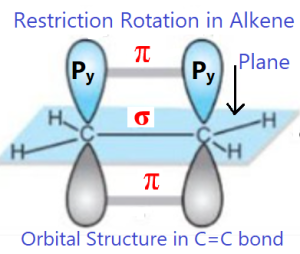
In alkene molecule, the sigma (σ) bond is formed by the overlap of sp2 hybrid orbitals; on the other hand, the pi (π) bond is formed by the overlap of unhybridized p-orbitals. The two carbon atoms of the C=C bond and the four atoms that are attached to them lie in one plane and their position in space are fixed. Rotation around the carbon-carbon double (C=C) is not possible, as rotation will break the pi (π) bond. So the main reason cis-trans isomers in alkene is the resistance of rotation in around of C=C bond.
The same case is occurred in cyclic alkane. Here, any rotation is not possible in accordance to C-C bonds which may form a ring. For rotation, C-C bonds and ring may be broken.
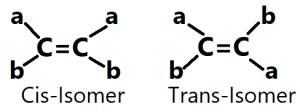
Classification of Geometric Isomers: These isomers are of two types; (i)Cis-isomer and (ii)Trans-isomer.
(i)Cis-isomer: The prefixes ‘cis’ means on this side of molecule. So, cis indicates that the groups are on the same side of the carbon-carbon double bond and ring in the molecule. The groups show strong repulsive force and strong steric hindrance. The intermolecular force is decreased and the molecules cannot arrange to form crystals. So, they have lower melting point.
(ii)Trans-isomer: The prefixes ‘trans’ means across. Therefore, trans indicates that the groups are on the opposite sides of the carbon-carbon double bond and ring in the molecule. The groups are far apart and there exists no repulsive force and steric hindrance between the groups. The intermolecular force is increased and the molecules can arrange to form crystal lattice. So, they have higher melting point.
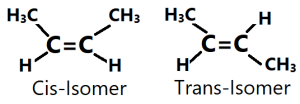
Examples of Cis-Trans-isomers:
(b)Optical Isomers and Optical Isomerism
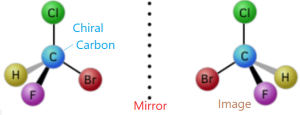
Optical isomer is a type of isomer showing by two organic compounds each of which has a chiral carbon or an asymmetric carbon atom which is bonded to four different radicals or groups or atoms. Optically active substances can form optical isomers. An optically active
compound is one that rotates the plane polarized light. Plane polarized light is the light which vibrates in only one plane. The optical isomers can rotate the plane polarized light in opposite directions when passed through them. The optical rotatory powers of two isomers are equal in magnitude but opposite in sign. These isomers are two types: (i) Dextrorotatory and (ii) Levorotatory optical isomers.
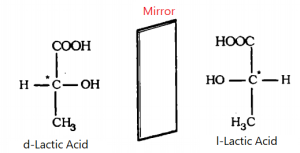
(i) Dextrorotatory optical isomers: The Latin word ‘dexter’ means ‘right’. The isomer which rotates the plane of polarized light to the right (clock wise direction) is known as dextrorotatory isomer or ‘d’-isomer or ‘+’-isomer.
(ii) Levorotatory optical isomers: The Latin word ‘laevus’ means ‘left’. The isomer which rotates the plane of polarized light to the left (anti clock wise direction) is known as levorotatory isomer or ‘l’-isomer or ‘-’-isomer.
Determination of specific rotation of optical isomer:
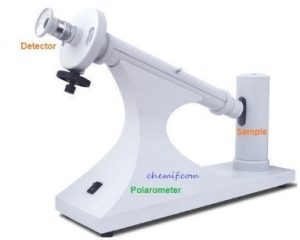
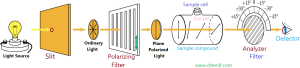
Specific rotation is the number of degrees of rotation of plane polarized light observed by the compound being examined present to the extent of 1g/mL taken in a 1 decimeter tube. A polarimeter (please see the figure) is used to detect optical activity as well as measure the specific rotation. It consists of a light source, two polaroid or nicol lenses and a tube of 1 dm length between the lenses two to hold the substance that is being examined to detect the optical activity. These components are arranged so that the light can pass through one of the lenses (polarizer) , then the tube, the second lens (analyzer) and finally can reach our eye.
When the tube is empty, the maximum amount of light reaches our eye when the two lenses are so arranged that they pass light vibrating in the same plane. If we rotate the lens that is nearer our eye, we find that the light dims, and reaches a minimum when the lens is at right angles to its previous position.
To test the optical activity and measure the specific rotation, a definite amount (1g/mL) of sample compound is taken in a sample cell. Then, ordinary light from light source is passed through a slit and this light is converted to plane polarized light which is then passed though the sample cell as well as sample. If we get maximum light from detector, the compound is said to be optically inactive. On the other hand, if we get maximum light by rotating or adjusting the analyzer, then the sample is optically active. If the rotation of the plane, and hence rotation of the analyzer is to the right(clockwise) , the sample compound is dextrorotatory (Latin: dexter means right) ; if the rotation is to be left (counterclockwise), the sample is levorotatory (Latin: laevus means left).
Using polarimeter, we can determine not only the direction of rotation but also the amount of rotation in degrees from the reading of analyzer as it is scaling with degrees. The symbols + or d is and – or l are used to indicate rotations to the right and to the left, respectively.
As for example, the lactic acid which is extracted from muscle tissue rotates plane polarized light to the right, so it is known as d- lactic acid or (+)-lactic acid. The 2-methyl 1-butanol which is obtained from fusel oil rotates plain polarized light to the left, so it is known as l-2-methyl 1-butanol or (-)-2-methyl 1-butanol.
Various types of Optical Isomers:
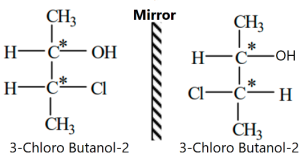
Enantiomer:
The stereoisomers that are mirror images of each other are called Enantiomers. In chemistry, an enantiomer is one of two stereoisomers that are mirror images of each other that are non-superimposable, much as one’s left and right hands are mirror images of each other that cannot appear identical simply by reorientation.
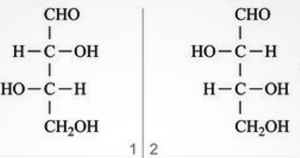
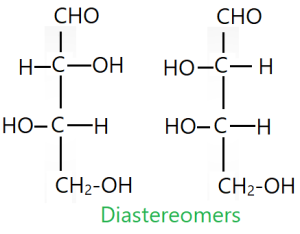
Diasteriomers:
Diastereomers are a type of a stereoisomers that are not mirror images of each other.. Diastereomers are defined as non-mirror image non-identical stereoisomers. Hence, they occur when two or more stereoisomers of a compound have different configurations at one or more of the equivalent stereo centers and are not mirror images of each other.
Racemic Mixture:
In chemistry, a racemic mixture, or racemate, is one that has equal amounts of left- and right-handed enantiomers of a chiral molecule. The first known racemic mixture was racemic acid, which Louis Pasteur found to be a mixture of the two enantiomeric isomers of tartaric acid.
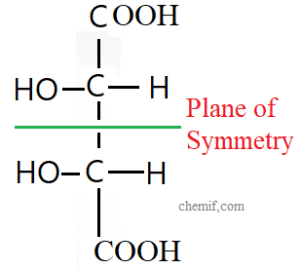
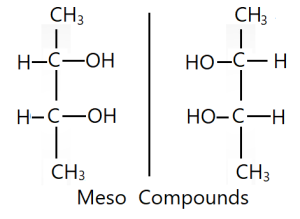
Meso compounds or Mesomers
A compound which is optically inactive although having two chiral centers is known as a meso compound. A meso compound has an internal plane of symmetry that separates its structure into two similar chiral centers. Specific rotation value of one half of this compound with one chiral center is equal to that of second half of this same compound
with second chiral center but opposite in directions. The mirror image of one half of meso compound is superimposable on that of second half. As a result, total specific rotation values of plane polarized light of the meso molecule becomes zero. So it is optically inactive. A meso compound or meso isomer is a non-optically active member of a set of stereoisomers, at least two of which are optically active. A meso compound is a stereoisomer with two or more chiral centers but no optical activity due to an internal plane of symmetry. In other hand, mesomers are organic molecules that have two chiral carbons that are identical.
This means that despite containing two or more stereogenic centers, the molecule is not chiral. A meso compound is “superimposable” on its mirror image. Example: meso-2,3-Buta-di-ol.