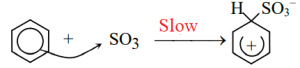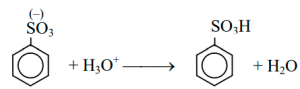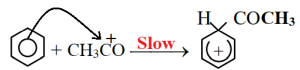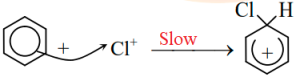Mechanism of Electrophilic Substitution Reaction in Benzene
Introduction: A chemical reaction in which a functional group or a radical is replaced by another functional group or radical is called the substituted reaction. Electrophilic Substitution Reaction of benzene is the reaction where an electrophile substitutes the hydrogen atom from a benzene ring. Basic examples of electrophilic substitution reaction of benzene are nitration, sulfonation, halogenation, Friedel Craft’s alkylation and Friedel Craft’s acylation reaction. For example, in nitration reaction, nitrobenzene is formed by reaction of benzene and concentrated nitric acid and sulfuric acid-
![]()
Mechanism of Electrophilic Substitution Reaction in Benzene:
A characteristic reaction of aromatic compound (Benzene) is electrophilic substitution reaction. A two-step mechanism has been proposed for these electrophilic substitution reactions. In the first, slow or rate-determining, step the electrophile forms a sigma-bond to the benzene ring, generating a positively charged benzenonium intermediate (sigma complex). In the second, fast step, a proton is removed from this intermediate, yielding a substituted benzene ring
1. Generation of electrophile: Due to the presence of Lewis acid, generation of electrophile takes place. As the Lewis acid accepts the electron pair from the attacking reagent.
2. Formation of arenium ion: The electrophile (E+) generated attacks on the benzene ring to form positively charged cyclohexadienyl cation called an arenium ion (sigma complex) containing one sp3 hybridized carbon atom. The positive charge is effectively distributed over three carbon atoms by resonance which makes it partially stable.
![]()
As the delocalization of electron stops at an sp3 hybridized carbon atom, the arenium ion is not aromatic in nature.
3. Removal of positive charge from the carbocation intermediate: The arenium ion finally loses its proton from sp3 hybridized carbon to a Lewis base restoring the aromaticity
![]()
Nitration Reaction:
Benzene reacts with a mixture of concentrated nitric and sulfuric acids to create nitrobenzene.
![]()
Mechanism:
Sulfuric acid ionizes to produce a proton. Nitric acid accepts the proton in an acid‐base reaction.
The protonated nitric acid dissociates to form a nitronium ion ( +NO 2).
![]()
The nitronium ion acts as an electrophile and is attracted to the π electron system of the benzene ring. The nonaromatic carbocation that forms has its charge delocalized around the ring.
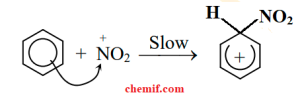
Electrophilic attraction The aromaticity of the ring is reestablished by the loss of a proton from the carbon to which the nitrogroup is attached and nitrobenzene is produced.
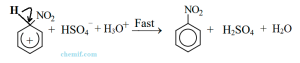
Formation of Nitrobenzene
Sulfonation:
The reaction of benzene with concentrated sulfuric acid at room temperature produces benzenesulfonic acid.

Sulfonation reaction
Mechanism:
The sulfuric acid reacts with itself to form sulfur trioxide, the electrophile. The sulfur trioxide is attracted to the π electron system of the benzene molecule.
sulfur trioxide formation
Electrophilic attraction the aromaticity of the ring is reestablished by the loss of a proton from the carbon to which the sulfonate group is attached and benzene sulfonic acid is produced.
The charge around the ring is delocalized, and then the loss of a proton reestablishes the aromaticity of the ring.
Friedel Craft Alkylation Reaction:
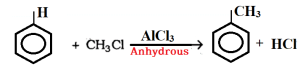
In presence of anhydrous Aluminium chloride (AlCl3) benzene reacts with alkyl halide (e.g. methyl chloride ) to produce alkyl benzene (e.g. toluene).
For example:
Mechanism:
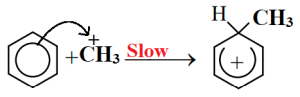
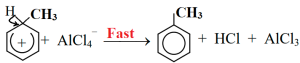
At first methyl chloride reacts with anhydrous AlCl3 and forms electrophile carbocation.
Carbocation forms sigma complex combining with benzene ring.
Then by elimination of proton from benzene sigma complex toluene is formed.
Friedel Craft Acylation Reaction:
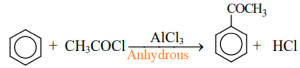
In presence of anhydrous Aluminium chloride (AlCl3) benzene reacts with acetyl chloride (acyl halide) to form acetyl benzene (acetyl benzene).
For example:
Mechanism:
At first acetyl chloride reacts with anhydrous AlCl3 and forms electrophile carbocation.
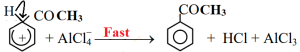
Carbocation forms sigma complex combining with benzene ring.
Then by elimination of proton from benzene sigma complex acetophenone is formed.
Chlorination Reaction:
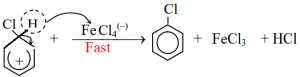
In presence of anhydrous ferric chloride, chlorine gas reacts with benzene and produces chlorobenzene.
Mechanism:
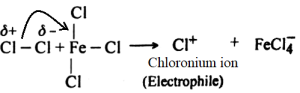
At first chlorine reacts with anhydrous FeCl3 and forms electrophile, chloronium ion.
Chloronium ion forms sigma complex combining with benzene ring.
Then by elimination of proton from benzene sigma complex, chlorobenzene is formed.